 |

Search

|
 |
 |
Eastern equine encephalomyelitis
|
 |
Eastern equine encephalomyelitis (EEE) is an arthropod-borne disease belonging to the genus Alphavirus. It causes severe and sometimes fatal encephalitis in game birds and various mammalian species (e.g. horses and humans). The disease was firstrecognized in 1831 when 75 horses died of an encephalitic condition in Massachusetts. The EEE virus was isolated in 1933 when a major outbreak of encephalomyelitis occurred in horses in the coastal areas of Delaware, Maryland, New Jersey, and Virginia.
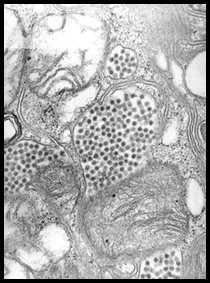
Eastern equine encephalomyelitis is a single-stranded, enveloped RNA virus in the family Togaviridae. The genus Alphavirus contains two other viruses, Western equine encephalomyelitis (WEE) and Venezuelan equine encephalomyelitis (VEE), which are closely related to EEE. |
Eastern equine encephalo-myelitis is endemic in Central America, the eastern part of South America, the Caribbean, and regions of North America. Two antigenic variants of EEE are recognized and are geographically categorized. The more virulent North American variant is found in the eastern United States, Canada, and the Caribbean and seems more likely to infect humans. The SouthAmerican variant is found in South and CentralAmerica. In the United States, EEE is most prevalent in the Atlantic and Gulf Coast states, but occasional outbreaks have been identified in Texas, Minnesota, and South Dakota, as well as in most states east of the Mississippi River. In the northern regions of the United States, EEE cases occur in the late summer to early fall in and around lowland swamp areas.
Small species of passerine birds are the principal vertebrate reservoir for EEE. They rarely become ill with the disease but serve to maintain and amplify the virus. The enzootic vector responsible for transmitting the virus among passerine, poultry, and game birds is the Culiseta melanura mosquito. This species of mosquitoes is ornithophilic, so it is less likely to play a role in the transmission to horses and humans. Theepizootic mosquito vectors, or bridging vectors are believed to be Coquillettidia spp. and Aedes spp. These species ofmosquitoes pick up the virus from infected birds and then infect mammals. Horses and humans are regarded as accidental or dead end hosts because they do not develop a sufficiently high viremia to re-infect mosquitoes and maintain the transmission cycle. |
| After initial infection with EEE, the virus replicates in myocytes, fibroblasts, reticuloendothelial cells,lymphoid cells and osteoblasts. One study found that active osteoblasts may be one of the main sites of virus amplification. This could explain why young animals are more susceptible to serious infection and develop higher levels of viremia. Following initial amplification, there is a primary viremia, and replication occurs in other extraneuronal tissues (e.g. lymph nodes andmuscles) prior to the second viremic phase.Eventually, through a hematogenous route, EEE is thought to enter the central nervous system (CNS). Once in the CNS, viral replication is believed to occur in neurons, vascular endothelial cells, and glial cells. Eastern equine encephalomyelitis is thought to induce damage by stimulating apoptosis of neuronal and glial cells. The incubation period of EEE is 4-10 days. The initial clinical signs of EEE in affected horses caninclude any of the following: biphasic fever, anorexia, somnolence, head pressing, stiff neck, and proprio-ceptive deficits. |
|
|
These signs can progress rapidly tocortical blindness, circling, head tilt, nystagmus, strabismus, paralysis, seizures, and possibly death. Some horses show no clinical signs before dying. The case fatality rate is 50-90% in horses and 30-50% in humans. Animals that recover commonly have mild to moderate long-term neurological dysfunction.
The clinical signs of EEE are indistinguishable from WEE, VEE, and other encephalitides, such as rabies, leukoencephalomyelitis, equine herpes virus 1, equine protozoal myeloencephalitis, and West Nile virus. Therefore, additional antemortem or postmortem tests are required for a definitive diagnosis. The ante-mortem diagnosis is based on detection of a single serum high titer or an elevation in paired titers. It can be difficult to get paired titers because horsescommonly will not live long enough to be able to get a second serum sample, Detection of IgM in the early stage of infection is made by antigen capturing ELISA. In addition, there are other serological tests available such as hemagglutination inhibition, complementfixation, and virus neutralization assay. A definitive diagnosis often is made postmortem with immuno-histochemistry, virus isolation, or reverse transcription polymerase chain reaction (RT-PCR) on nervoustissues.
Usually, no gross lesions are seen in the brain or spinal cord at necropsy; however, occasionally, hyperemia, congestion, and focal areas of hemorrhage are found in the gray matter. A characteristic change to differentiate EEE from other encephalitides is diffuse lymphocytic/neutrophilic perivascular cuffing andinfiltration of the gray matter of the cerebral cortex, the thalamus, and hypothalamus. Eastern equineencephalomyelitis infected animals can die so acutely that neutrophils are still the predominant inflammatory cell type. The cerebellum is often affected less severely, and only mild changes are observed in the gray matter of the spinal cord. Less definitive findings are endothelial cells swelling with prominent hyaline thrombi, perivascular hemorrhage and edema, diffuse microgliosis, cortical necrosis, and neuronal degeneration.
There is no specific treatment for EEE.Treatment is generally focused on supportive care, which includes providing padding to protect animals from self-induced trauma, anti-inflammatory drugs to limit the secondary inflammation, fluids and electrolytes. More important is prevention of EEE infection. There are several inactivated monovalent, bivalent (EEE, WEE) andtrivalent (EEE, WEE, VEE) vaccines available. It is recommended to give three vaccinations tounvaccinated horses and foals 4-6 weeks apart. Then give a booster vaccination annually prior to peakmosquito season. A booster vaccination can be given to mares 4-6 weeks prior to foaling to increase the concentration of antibodies in their colostrum. Foals should be given their first vaccination at approximately 6 months of age or, if the mare was unvaccinated, at 3-4 months of age.
Controlling vectors, elimination of breeding sites and control of mature insects will increase the success of preventive measures, particularly during outbreaks of disease. Horses may be partially protected frominsects if repellant is used.
In late summer and early fall of 2007, several horses in Indiana were diagnosed with EEE infections. Initial diagnosis was made at Purdue ADDL and later confirmed at NVSL by PCR and/or VN, IgM capturing ELISA. No human fatality has been recorded due to EEE virus infection in Indiana.
-by Chad Frank, Michigan State University SVM Extern
-edited by Dr. Roman Pogranichniy, ADDL Virologist
References:
-
Animal and Plant Health Inspection Service web site. Eastern Equine Encephalomyelitis. Available at: www.aphis.usda.gov/pa/pubs/fsheet_faq_notice/fs_aheasterne.htm. Accessed September 23, 2007.
-
Murphy AF., Gibbs PJ, Horzinek MC et al: 1999. Flaviviridae. Veterinary Virology 3rd ed. San Diego CA: Academic Press. Pp 550--553.
-
Calisher CH: 1994. Medically Important Arbo-viruses of the United States and Canada. Clinical Microbiology Reviews: 89-116.
-
Brault AC, Powers AM, Chavez CL et al: 1999.Genetic and Antigenic Diversity Among Eastern Equine Encephalitis Virus from North, Central, and South America. American Journal of Tropical Medicine and Hygiene. 61: 579-586.
-
Saif YM: 2003. Other Viral Infections. Diseases of Poultry, 11th ed. Ames, IA: Iowa State Press.Pp 388-392.
-
Vogel P, Kell WM, Fritz EL et al: 2005. Early Events in the Pathogenesis of Eastern Equine Encephalitis Virus in Mice. American Journal of Pathology. 166: 159-171.
-
Jubb KFV, Kennedy, Palmer N: 1993. Inflammation of the Central Nervous System. Pathology of Domestic Animals, 4th ed. San Diego CA: Academic Press. Pp 412-414.
-
Smith BP: 2002. Nervous System. MerckVeterinary Manual, 9th ed. Whitehouse Station NJ: Merck & Co., Inc. Pp 1031-1032.
-
Kahn CM: 2002. Nervous System. Merck Veterinary Manual. 9th ed. WhitehouseStation, NJ: Merck & Co., Inc. Pp 1031-1032.
-
Piero FD, Wilkins PA, Dubovi EJ et al: 2001.Clinical, Pathological, Immunohistochemical, and Virological Findings of Eastern Equine Encephalomyelitis in Two Horses. Veterinary Pathology 38: 451-456.
|
|
 |

|
 |